Solid-state batteries: the new frontier of electrification?
9 March 2022
The lithium battery sector is constantly evolving, with real research every day into developing increasingly high-performance, innovative technologies, which can guarantee greater range, power and ever shorter charging times.
Solid-state battery technology may seem like the final frontier of technology in this sense, a solution being finalised that has all the potential to become the future of electric mobility.
Listeners to Battery Weekly know this all too well, the live weekly programme on the Flash Battery LinkedIn and YouTube channels, where every Monday at 6:00 pm, our experts, Marco Righi, Alan Pastorelli and Daniele Invernizzi review the latest news from the world of electrification and where the subject of solid-state batteries has become one of the hottest topics.
But when we talk about solid state, a variety of different factors come into play, with a series of great advantages but also many limits that are still being studied and, to date, are delaying its entry onto the market. Let’s clear things up.
Solid-state batteries
What are solid-state batteries and how do they work: differences with lithium batteries
A solid-state battery is essentially battery technology that uses a solid electrolyte instead of liquid electrolytes which are instead behind lithium-ion technology.
To be able to talk clearly about solid-state batteries, it is therefore important to take a step back and understand how lithium-ion batteries work in detail and their main differences compared with this new technology.
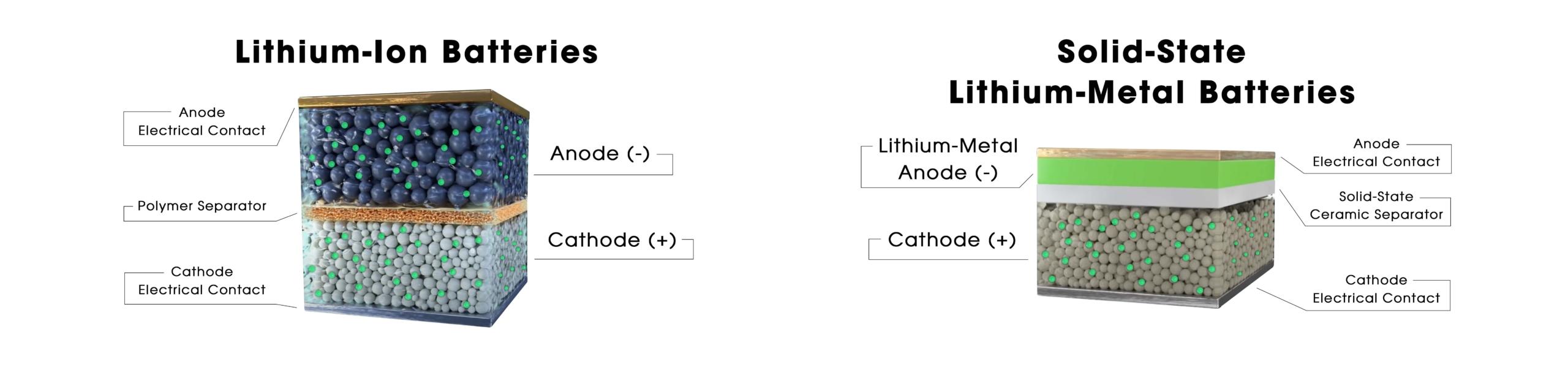
The structure of a lithium-ion battery
The following picture shows the structure of an actual lithium-ion cell, the technology that is now used in the vast majority of electric vehicles in circulation. What are we looking at?


Every lithium-ion cell has:
- Two electrodes, i.e. compounds that can accept the intercalation of lithium ions inside their structure. More specifically, we are talking about:
- A cathode, i.e. the positive pole of the battery made of cathodic material (eg. LFP, NMC, LMO, etc) and the current collector
- An anode, i.e. the negative pole of the battery made of anodic material (eg. carbon or graphite) and the current collector
- A central separator, i.e. a thin layer made from a plastic polymer (polyethylene or polypropylene) which acts as a mechanical separator between the anode and cathode and works as an insulator.
- An electrolyte: i.e. the medium through which ions move; an organic liquid that contains lithium salt. The electrolyte fills the entire volume inside the cell, soaks the electrodes and allows the lithium ions to move by acting as a connecting link between the cathode and anode.
In a current lithium-ion battery, the separator does not have any other functions apart from insulation and is totally submerged in the liquid electrolyte which soaks everything inside the cell and becomes a real medium through which lithium ions move between the cathode and anode, where the anode is made from a graphite structure. The lithium ions therefore move through the electrolyte and intercalate in the crystal structures of the two anode and cathode electrodes (structures which have empty spaces inside, where the lithium ions fit as they are very small particles).
The structure of a solid-state battery
However, the internal structure of a solid-state cell is very different, as all its parts are solid. While in traditional lithium batteries, the electrolyte is a liquid, solid-state cells are formed of:
- A cathode (or positive electrode), which can be made with the same compounds as a lithium-ion battery (eg. LFP, NMC, LMO, etc)
- A separator, generally ceramic or solid polymer, which also works as the electrolyte
- An anode made of lithium metal (pure lithium)


The grey central layer is the solid-state separator which, on its own, acts both as the separator between the anode and cathode and as the electrolyte. It therefore becomes the medium through which the ions move and also has electric insulating properties and as a mechanical separator between the anode and cathode. The fact that there is this solid, resistant support allows the removal of the graphite structure on the anode part and ensures that lithium metal accumulates directly on the anode (there are also semi-solid solutions where the electrolyte is a gel).
How does a solid-state battery work?
When the cell is charging, the lithium particles move from the cathode, through the structure of the atoms that form the separator, and then move in between the separator itself and the anode’s electrical contact, thus forming a solid layer of pure lithium. In this way, the anode will only be formed of lithium particles and will have a smaller volume than a lithium-ion technology anode, which contains the graphite structure.
Do you know about Battery Weekly?
The LIVE weekly programme every Monday at 6:00 pm on the Flash Battery LinkedIn and YouTube channels discussing the latest electrification trends
Solid-state battery technology
What are the current strengths of solid-state battery technology
On paper, solid-state batteries promise many improvements over the current batteries on sale; in fact, solid electrolytes seem to offer greater energy density, a longer life and greater safety, all in a smaller size.
But it is important to remember that this technology is still in the development phase and, to date, lithium-ion batteries remain the best performing technology on sale, with a variety of chemistries, each used for different purposes, readily available and mass produced.
However, let’s have a look at the advantages offered by solid-state batteries:
The key factor of safety
Solid-state batteries do not have a liquid electrolyte, which in lithium-ion batteries is one of the most challenging components in terms of safety, because it is volatile and therefore more flammable. Furthermore, this is replaced by a thicker separator layer formed of a material that is mechanically more resistant to high temperatures (because it has a ceramic composition with various additives); this makes the separation between the anode and cathode more reliable, so much so that it prevents short circuits, even in the event of misuse or deterioration, and therefore the intrinsic safety of the cells increases.
Of course, not all lithium-ion batteries have the same level of safety. We spoke about this subject in the article “Risks associated with lithium: Can you really trust a lithium battery?”
Another advantage in terms of safety is the greater resistance to dendrites, or the sharp, uneven build-up of lithium that forms during movement from the cathode to the anode. In fact, lithium does not move evenly and tends to group together and form points which, like real pins, grow and, in some extreme cases, can pierce the separator. However, thanks to its thickness, solid separators are more resistant to piercing from dendrites and therefore avoid possible short circuits and the gradual deterioration of the cell.


Record energy density
The greater intrinsic safety helps bring another major improvement: the use of a pure metal anode encourages a huge increase in energy density. This is essentially down to the removal of the graphite anode (which in lithium-ion batteries contains the ions when they migrate). In a solid-state battery, during the transfer, only the ions remain and a bulky, heavy compound part is removed which does not actively help generate energy.
According to the latest studies, solid-state batteries have an energy density 2-2.5 times higher than current lithium-ion technology and this huge advantage would result in a lighter and smaller battery. This is certainly a breakthrough for electric mobility, which would benefit from greater range and a lighter weight, but let’s remember that we will only be certain of this figure when this technology is officially ready.
Ultra-fast charging times
The latest studies have shown that solid-state batteries are able to charge up to 6 times faster than the current technologies on sale. But this figure is also still uncertain and will depend on how this new technology is developed. There are already solid-state battery prototypes that charge very quickly, but to the detriment of other decisive factors for achieving good performance. We will need to weigh up this advantage with other essential characteristics that these batteries should have, only then can we assess the best alternative, including in terms of cost.
To date, what is certain is that liquid electrolytes tend to suffer at high temperatures, while solid electrolytes, on the contrary, become more high-performance at high temperatures and this would support their performance during fast charging, an operating phase that typically produces much higher temperatures.
Quicker production
Some people argue that a solid-state electrolyte, as it is not liquid, can allow a quicker, easier production process, which uses less material and energy; but this theory, while understandable, also cannot yet be proven and only will be when this technology is truly mass produced.
However, we can certainly say that at the moment the filling of the cell with the electrolyte is a process that requires plenty of time: the cell must be assembled empty and it must have a hole so the electrolyte can be filled up later on, you will then have to wait for the electrolyte to be completely absorbed and, afterwards, you will need to refill it to bring it to the right level and seal it. It therefore is certainly an influential phase in the production process and, with solid-state technology, there could potentially be a real improvement, but in order to draw sound conclusions, we need to wait for actual production of this type of cells.
Main fields of application
But what will be the main fields of application of solid-state batteries?
As we have seen, solid-state batteries of the near future will potentially be able to provide huge advantages that will increase the performance and efficiency of vehicles and will revolutionise the electrification sector of the automotive industry. But the arrival of solid-state technology onto the market already seemed imminent a few years ago and instead the breakthrough has still not happened. How come?
Just as there are many advantages, there are also certain limits due to how young this technology is, as it is still not ready and constantly evolving. This is why we can call these limits real challenges to be addressed and major new goals to be achieved. Let’s find out together.
Stability problems
During charging and discharging, it is though the solid-state cell is breathing. The thickness of the lithium-metal anode increases during charging and decreases during discharging, and just like all unstable elements, this will eventually cause deterioration.
The main problem comes from the difficulty of keeping the solid-state cells fixed and compressed at the same time.
A cell should be compressed so that the internal layers do not detach, but it is not enough to fasten it to a containing structure, because this will constantly need to “breathe”. You therefore need to create a complex mechanical structure: in “tabletop” solid-state battery prototypes, plates are installed with springs that keep everything compressed, but this is a complex and expensive system that cannot be mass produced.
Due to its composition, it is not possible to stop a solid-state cell from swelling; however, research can work on how to make it less demanding in terms of pressure (so that the cell remains stable on its own without needing all this pressure, but perhaps only with the use of a filler), or on the study of advanced materials that allow the cell to expand while keeping it firmly fixed and compressed.
The separator only performs at a high temperature
Ions are matter, atoms, and it therefore makes sense that they move more easily in a liquid, while a solid (a ceramic separator) must have a special composition to be able to allow ions to move freely.
There are already high-performance separators in this sense, but only at high temperatures, because solid electrodes only become good conductors at temperatures above 50 degrees. This limit means that solid-state technology is still hardly used in real vehicles, because we cannot assume that the battery is always hot. When the solid-state battery is not hot, its performance currently falls considerably. Work will need to be done to ensure that the solid electrolyte performs well at increasingly lower temperatures.
Life-cycles are still short
The life-cycles of solid-state batteries currently being tested are still shorter than other lithium-ion technologies, such as LFP chemistry, which easily surpasses 4,000 charge cycles.
The main problem is the fact that it is very difficult to get good contact between all the cell’s layers. When you start to lose contact between the layers, the cell loses capacity and performance.
High cost
The cost of a solid-state battery is currently very high because we are talking about an extremely innovative technology; so the costs of both the materials and the production processes need to be higher than for mass-produced batteries. It is not yet clear what the final cost of this technology could be, but we can certainly assume that, if major car manufacturers are investing in this direction, they have enough evidence to believe that the cost can also be adapted to mass production.
Application
But what will be the main fields of application of solid-state batteries?
Even though solid-state batteries still have a few problems to be resolved, their arrival on the market is now certain and we can expect their widespread use in any sectors where, to date, energy density is a limiting factor, because the space is currently not enough to store all the energy that is needed. In fact, as they have twice the energy density, solid-state batteries will double the range and are now seen as the future of the automotive market and, more generally speaking, of all transportation.
The industrial machinery sector and the electric vehicle sector are also looking at this new technology with interest: this is the case for very energy-intensive machinery or heavy vehicles, which often require extensive range and where, to date, the volume is low compared with the amount of energy that could be used.
The introduction of solid-state battery technology could definitely be useful for further expanding the category of electrified vehicles. If, along with their large energy density, solid-state cells therefore became competitive on all fronts, they could undoubtedly be a valid path for the future of industrial electrification too.
“There is much promise and high expectations about the development of solid-state batteries which, once perfected, could be the key to bringing electrification to more challenging sectors too, which currently cannot yet abandon fossil fuels due to energy density limits. Our Research and Development department is certainly looking at this new technology with interest, but it is important to remember that innovation is a constantly evolving concept, which should not be followed but rather anticipated. Here at Flash Battery, we test and study new materials and methodologies every day with an increasing focus on high efficiency and sustainability, with the goal of making industrial electrification increasingly widespread and open to the most diverse sectors”.
Alan Pastorelli
CTO and Co-Founder of Flash Battery – LinkedIn
Electric market
When will solid-state batteries arrive on the electric market?
Solid-state batteries are not science fiction, on the contrary! They are already reality in small applications, such as certain consumer batteries or some vehicles like buses, suitable for intensive use and where the battery is used non-stop for the whole day and even though it stays hot, it works without too many problems.
Solid-state technology is therefore already used with low volumes in:
- Batteries that work in controlled climate conditions
- Batteries for aerospace applications
- Semi-solid or solid-state hybrid batteries
One real life example are the 50 E70 vehicles with semi-solid-state batteries recently launched on the market by the Chinese Dongfeng Motor Corporation: it is a sort of technological first, where it seems that semi-solid-state batteries have shown excellent electrochemical properties through a series of simulation tests.
How will they behave in the long-term? The eyes of the entire automotive world are certainly on this Chinese car manufacturer, so we will see how it goes. It could also just be a great marketing opportunity to promote their brand and associate it with this new technology before the others, but there is no guarantee that they will reach mass production.
What is certain is that real solid-state batteries for automotive use are still in an experimental stage, with major challenges that are still ongoing and, to date, are limiting their mass production. However, many car manufacturers are interested in this promising technology, such as Mercedes, Volkswagen, Toyota and many others, which are investing huge resources into studying and developing it. They will be the ones to have the first definitive technology, already announced for between 2024 and 2026, provided the limits are resolved.
Would you like to stay up-to-date about all the latest news in batteries and electrification?
Don’t miss Battery Weekly, every Monday LIVE at 6:00 pm on the Flash Battery LinkedIn and YouTube channels
Notes
Source Fig. 1: image inspired by the Quantum Scape video “What Are Solid-State Lithium-Metal Batteries?” https://youtu.be/azACL3lLMo8
Source Fig. 2: image inspired by the Quantum Scape video “What Are Solid-State Lithium-Metal Batteries?” https://youtu.be/azACL3lLMo8
Source Fig. 3: image taken from the article “Source of Detrimental Dendrite Growth in Lithium Batteries discovered” – MSE Supplies https://www.msesupplies.com/blogs/news/source-of-detrimental-dendrite-growth-in-lithium-batteries-discovered
Our Blog
Would you like to talk to us about your electrification project?